A commentary on how Neurons fire in Early Alzheimer’s: Linking Electrophysiology to Microglial States
A few days ago the Alzforum covered again my recent work and its relation to ongoing work in the field of immunology after news emerged following the Alzheimer’s Association International Conference in Toronto
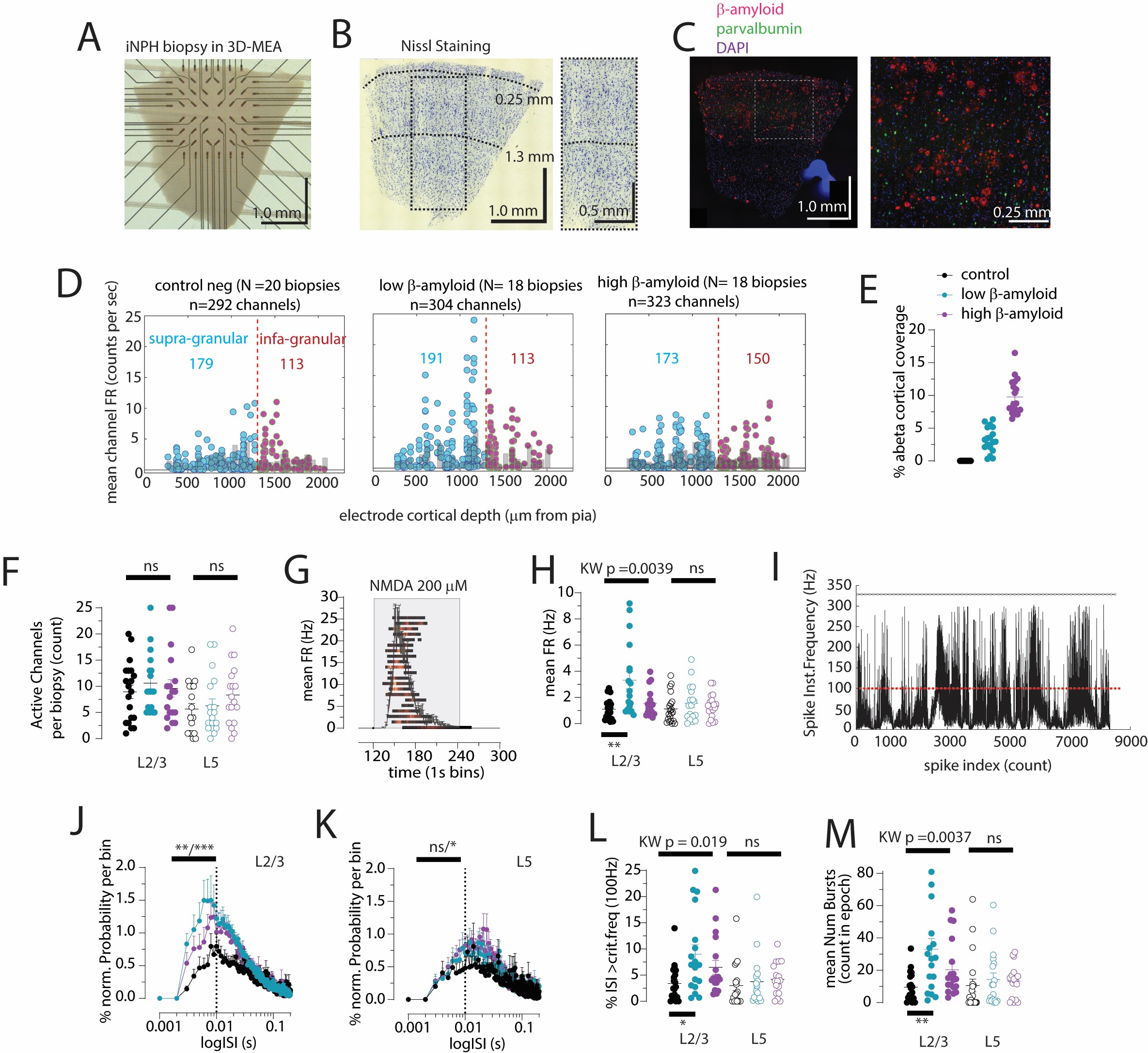
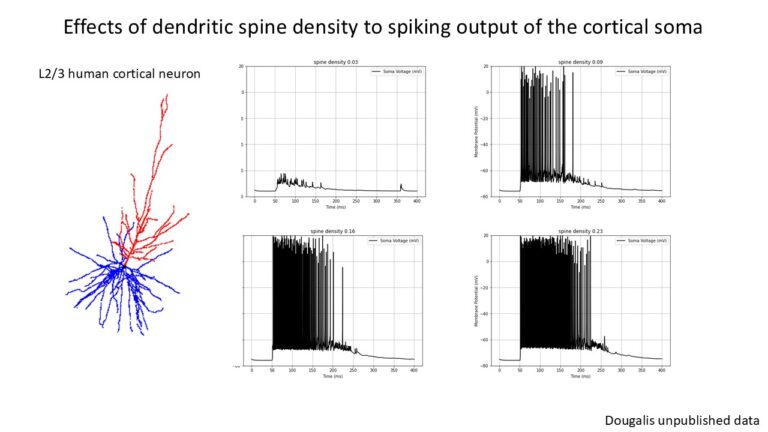
My latest work (Dougalis et al., 2025, https://www.biorxiv.org/content/10.1101/2025.01.07.630516v1.full) is diving into the electrophysiological consequences of early Alzheimer’s pathology in human neurons—and the findings are striking. By recording from layer 2/3 pyramidal neurons in iNPH biopsy tissue, we’ve shown that amyloid and tau pathology reshape the electrical behavior of these cells while inhibitory control from layer 1 neurons falters. This gives a direct window into how pathology disrupts neuronal network function.
At the recent Alzheimer’s Association International Conference in Toronto, researchers from the iNPH cohort highlighted complementary insights:
Disease-associated microglia (DAMs):
The balance between DAM-like and homeostatic microglia predicts long-term cognitive decline. High DAM levels correlate with vulnerability, while homeostatic microglia appear protective.
Rare CSF macrophages: Newly identified macrophages in cerebrospinal fluid carry transcriptional signatures strongly linked to AD risk, suggesting immune surveillance in CSF may play a role in disease progression.
By combining electrophysiology with single-cell genomics, we can start connecting the dots: the neurons that misfire in the presence of amyloid and tau are the same ones exposed to shifts in microglial states. Understanding these interactions opens a new avenue for early detection and intervention.
The big picture is clear: living human tissue provides a rare opportunity to link molecular changes, cellular behavior, and cognitive outcomes. Our work shows that neuronal firing changes isn’t just a downstream symptom—it’s a measurable, mechanistic effect of early AD pathology, potentially guided by the surrounding immune environment.